News
Search EMA's news announcements
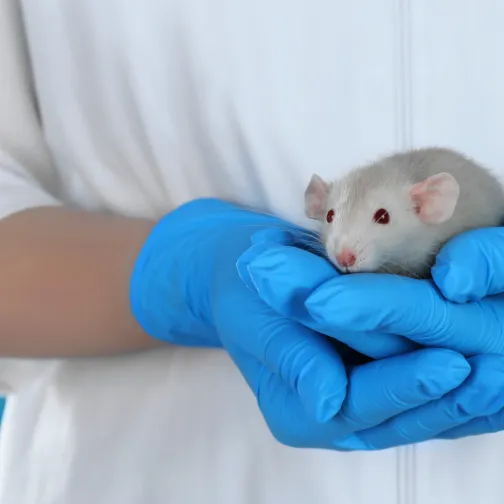
A draft qualification opinion for the new testing method is open for consultation
The European Medicines Agency's (EMA) office is closed from 18:00 on 1 April to 08:30 on 7 April
Annual updates to virus strains ensure vaccines stay effective
Imdylltra addresses unmet need in people with poor prognosis and limited treatment options

Five new medicines recommended for approval; another 13 medicines recommended for extension of their therapeutic indications
The medicine was not effective for the treatment of mpox in randomised clinical trials

Three new PRIME features streamline scientific dialogue and help developers to stay on track

Board adopts Agency’s 2025 annual report and notes electronic product information implementation roadmap
Outcomes of the Committee for Veterinary Medicinal Products (CVMP) meeting

PRAC warns about known risk of aseptic meningitis with chikungunya vaccine Ixchiq