Questions and answers: Article 31 pharmacovigilance referrals
HumanRegulatory and procedural guidanceReferrals
This page lists questions which stakeholders, in particular marketing authorisation holders (MAHs), may have on an Article 31 referral resulting from the evaluation of data from pharmacovigilance activities.
It provides an overview of the European Medicines Agency's practical and operational aspects with regards to the handling of Article 31 pharmacovigilance referral procedures. Revised topics are marked 'New' or 'Rev.' on publication.
A PDF version of these questions and answers is available below:
These questions and answers are for guidance only, without prejudice to legal and regulatory interpretation that might be provided in future updates of the rules governing medicinal products in the European Union, volume 2, notice to applicants. These questions and answers should be read in conjunction with the Directive 2001/83/EC of the European Parliament and of the Council of 6 November 2001 on the Community code relating to medicinal products for human use. Marketing authorisation holders (MAHs)/applicants must in all cases comply with the requirement of EU legislation.
Links to documents referred to in the below questions and answers can be found under Referral procedures: regulatory and procedural guidance.
An Article 31 pharmacovigilance referral follows the provisions of Article 31 of Directive 2001/83/EC.
It applies where the interests of the Union are involved, and only when the procedure is initiated as a result of the evaluation of data relating to pharmacovigilance of an authorised medicinal product(s)1.
The procedure for an Article 31 pharmacovigilance referral is laid down in Article 32, and Articles 107j(2) to 107k of Directive 2001/83/EC.
References
1When the procedure is initiated as a result of the from the evaluation of data relating to quality and/or efficacy of an authorised medicinal product(s), the procedure for an Article 31 non-pharmacovigilance referral will apply, and in such cases the matter should be referred to the Committee for Medicinal Products for Human Use (CHMP). Please refer to the Questions & Answers on the practical implementation of Article 31 non-pharmacovigilance referrals.
An Article 31 pharmacovigilance referral procedure should be initiated where the interests of the Union are involved and as a result of the evaluation of data relating to pharmacovigilance activities of an authorised medicinal product(s), and when none of the criteria listed in Article 107i1of Directive 2001/83/EC are met.
The term ‘interest of the Union’ refers particularly to the interests of public health related to medicinal products in the Union (for example in light of concerns related to the safety of a medicinal product) and to the free movement of products within the Union.
References
1Where one of the criteria listed in Article 107i(1) is met, the urgent union procedure laid down in Article 107ito 107k will apply, and in such cases the matter should be referred to the Pharmacovigilance Risk Assessment Committee (PRAC). Please refer to the .
An Article 31 pharmacovigilance referral can be initiated by the competent authorities in the Member States (MSs), the European Commission (EC) or by the marketing authorisation holder(s) (MAHs).
The initiator of the procedure refers the matter to the Pharmacovigilance Risk Assessment Committee (PRAC) by circulating a notification to the Agency, all MSs and the EC.
The notification will identify the safety concern and the question(s) referred to the PRAC for consideration. The notification will include a detailed explanation of the issue raised, and should address how the Union interests are involved.
Only the MS concerned or the EC can identify the question. Therefore in advance of initiating a referral under this Article, the MAH should contact a MS or the EC with a request to assess and confirm the Union interest. The MAH can include in the scope of the referral only its own medicinal product, with justification of potential extension to others.
The notification will be publicly available at the start of the procedure (please refer to Question 8).
A Member State (MS) may, where urgent action is necessary to protect public health, suspend the marketing authorisation at any stage of the procedure, and prohibit the use of the medicinal product(s) concerned on its territory until a definitive decision is adopted.
In this case, the MS informs the European Commission (EC), the Agency and all other MSs, no later than the following working day, of the reasons for its action.
All medicinal products affected by the safety concern referred and with a valid marketing authorisation (MA) in the European Economic Area (EEA) will be included in the Article 31 pharmacovigilance referral. This includes all medicinal products concerned by the safety issue, regardless of whether the MA was granted nationally (including via the mutual recognition and decentralised procedures) or via the centralised procedure. MA applications in the EEA may also be included. However, in case the safety issue concerns only centrally authorised medicinal products, the procedure under Article 20 of Regulation (EC) No 726/20041is initiated instead.
The referral procedure may concern a specific medicinal product, all medicinal products containing the same active substance (range of medicinal products) or all medicinal products belonging to the same therapeutic class (several active substances concerned).
The marketing authorisation holder(s)/applicants cannot choose whether or not to include their medicinal products in an Article 31 pharmacovigilance referral procedure. The inclusion of their medicinal products depends on the scope of the procedure that is defined by the safety concern raised in the notification.
Reference:
1 Please refer to the Questions & Answers on Article 20 Pharmacovigilance procedures.
The Agency, with input from the competent authorities of the Member States (MSs) within the European Economic Area (EEA) as necessary, will identify the authorised medicinal products concerned by the procedure. All authorised medicinal products concerned are identified using information from the Article 57 database. This will take place at the start of the procedure, and a draft list of the medicinal products identified will be publicly available on the Agency’s website on the specific procedure page (please refer to Question 8).
If it is concluded that the safety issue also concerns other medicinal product(s) (e.g. range of medicinal products, a therapeutic class) than the ones covered by the notification, the Agency can extend the scope of the procedure.
From a procedural view point, only those products identified at the start of the procedure will be covered by its scope. However, to the extent that other medicinal products affected by the concern but not identified at start of procedure are authorised in the EEA, or subject to future authorisations by the MSs, the concerned MSs should take due consideration of the scientific conclusions of the procedure and apply them to these products.
Upon receipt of the notification, the Agency, based on the information collected from the Article 57 database and possibly in consultation with the national competent authorities of the Member States (MSs) within the European Economic Area (EEA), will identify in which MS(s) the concerned medicinal product(s) is/are authorised.
If it is concluded that the scope of the procedure concerns medicinal product(s) authorised in only one MS, an Article 31 pharmacovigilance referral procedure will not be initiated, and the safety concern will be handled by the MS concerned.
Once the Article 31 pharmacovigilance procedure is triggered, the safety issue will be discussed at the upcoming Pharmacovigilance Risk Assessment Committee (PRAC) plenary meeting and a brief summary will be included in the agenda published at the beginning of the PRAC meeting.
The start of the procedure will be announced as part of the PRAC meeting highlights, which will be published on the next working day following the PRAC meeting during which the matter is considered.
The announcement will specify the safety issue under consideration and will include the publication of the following documents on the Agency’s website on a page created specifically for the procedure:
Reference:
Whenever possible, MAHs will be informed on the Wednesday before the PRAC meeting of new pharmacovigilance Article 31 procedure(s) that the PRAC will consider the following week. This communication will be provided for information only.
Following the PRAC meeting, a public announcement on the Agency’s website will include all information related to the start of procedure. In addition, all qualified persons for pharmacovigilance (QPPV) of the medicinal product(s) concerned by the Article 31 pharmacovigilance referral identified in the published product listing will be notified electronically (via e-mail/Eudralink) by the Agency. The notification of the procedure initiation to the QPPV will include:
The Agency may release updated information on the website during the procedure and therefore marketing authorisation holder(s) should continuously check the Agency’s website for any relevant updates (please refer to Question 29, Question 36 and Question 40).
The qualified person for pharmacovigilance (QPPV) will, by default, be the contact person and will receive all correspondence from the Agency regarding the Article 31 pharmacovigilance procedure.
The QPPV may if they wish to, either designate a different contact person within the organisation of the MAH or designate another party to represent the MAH for the procedure. In this case they must inform the EMA procedure assistant via email.
All documentation concerning the Article 31 pharmacovigilance referral will be sent to the contact person only. Receipt of any documents by the contact person will be considered to constitute effective receipt by the MAH inter alia for the purposes of calculating the procedural timelines.
All communications with the Agency should be channelled via the contact person only.
The marketing authorisation holders can form a group for the purpose of the procedure (irrespective of group/company affiliation) in order to provide a single consolidated response and/or oral explanations to the questions raised by the Pharmacovigilance Risk Assessment Committee (PRAC) during the procedure. In this case the cover letter accompanying the single consolidated response and/or request for oral explanation should clearly identify the parties responsible for the submission/request.
If a centrally authorised product is involved in an Article 31 pharmacovigilance referral, the qualified person for pharmacovigilance (QPPV) will be notified by the Agency of the start of the procedure as will all other QPPV (please refer to Question 9). The announcement on the Agency's website will also be linked to the EPAR page of the product.
The Agency will levy a fee for a pharmacovigilance referral under Article 31 of Directive 2001/83/EC.
The share payable by each marketing authorisation holder (MAH) will be calculated by the Agency based on information recorded in the Article 57 database. An advice note will be sent after the start of procedure, to the relevant qualified person(s) for pharmacovigilance (QPPV) in order to ensure the accurate identification of the chargeable units for the medicinal products involved in the procedure. Following the advice note, an invoice will be sent to each MAH.
For MAHs already qualified as a micro-, small or medium-sized enterprise (SME) by the Agency, or those that send a SME declaration within 30 days of the invoice date, the fee will be reduced (small- or medium-sized enterprise) or waived (micro-sized enterprise).
References:
The marketing authorisation holder(s) (MAHs) concerned by an Article 31 pharmacovigilance referral procedure will be requested to submit information relevant for the assessment of the safety concern.
The MAHs can present written or oral explanations to the Pharmacovigilance Risk Assessment Committee (PRAC) within the time limit as specified in the procedure timetable, and before a recommendation is issued by the PRAC.
For detailed information on how and when to submit data (please refer to Question 18 and Question 19).
Regardless of whether or not the MAHs present written or oral explanations to the PRAC, a recommendation applicable to all marketing authorisations concerned by the procedure will be issued by the PRAC.
The safety concern triggering the Article 31 pharmacovigilance referral will be substantiated by additional data that could be requested by the Pharmacovigilance Risk Assessment Committee (PRAC) in the format of a list of questions, comments on the scientific background supporting the triggering of the procedure or by using data sources available to the Agency and/or to the national competent authorities (NCAs) of the Member States.
This data may be gathered from several different sources (i.e. from concerned marketing authorisation holders (MAHs), healthcare professionals, patients’ organisations, Eudravigilance data, data available to the NCAs, etc.).
The need for specific data to be collected is identified by the PRAC at the start of the procedure.
The data to be considered for the assessment will have to be submitted within the specified deadline as published in the announcement of the start of the procedure (please refer to Question 8).
Notwithstanding the above, the PRAC may also collect additional data through further lists of outstanding issues, a public hearing and/or an oral explanation in accordance with an extended timetable, which will be made publicly available (please refer to Question 21 and Question 23).
The assessment of data within the Article 31 pharmacovigilance referral is led by the Pharmacovigilance Risk Assessment Committee (PRAC).
At the start of the procedure, the PRAC Chairperson appoints a PRAC rapporteur and PRAC co-rapporteur(s) who will perform the assessment of all data collected within the agreed timelines.
The assessment will result in the PRAC issuing a recommendation on the safety issue reviewed, which will be forwarded to the Committee for Medicinal Products for Human Use (CHMP) or to the Co?ordination Group for Mutual Recognition and Decentralised Procedures (CMDh), as applicable (please refer to Question 16).
Even though the assessment of the Article 31 pharmacovigilance referral will be performed by the PRAC, there will be a close collaboration during the assessment between the PRAC (co-)rapporteurs and the CHMP (co-)rapporteurs (in case at least one centrally authorised product is included in the scope of the procedure) or the CMDh member with the leading role (in case the concerned products are only nationally authorised including via the mutual recognition and decentralised procedures).
The Pharmacovigilance Risk Assessment Committee (PRAC) (co-)rapporteurs for an Article 31 pharmacovigilance referral should be appointed by the PRAC Chairperson from amongst its members or alternates (hereafter referred to as PRAC members).
The PRAC Chairperson will endeavour to apply the criteria of best available expertise to be taken into account for the appointment of the PRAC (co-)rapporteurs for each procedure.
In case of an Article 31 referral concerning several active substances belonging to the same therapeutic class, or where several issues are to be assessed, a lead rapporteur and several co-rapporteurs may be appointed.
References
Marketing authorisation holder(s) (MAHs) should submit to the Agency and all Pharmacovigilance Risk Assessment Committee (PRAC) members all available evidence to support the assessment of the impact of the safety concern being reviewed in the procedure in response to the list of questions and as per the timelines published on the Article 31 pharmacovigilance procedure page.
MAHs of medicinal products concerned by the procedure should submit their responses as follows:
Published data can be presented as supportive documentation in response to a specific question if no other data is available.
In case some questions (e.g. on a specific pharmaceutical form) are not applicable/relevant to all medicinal product(s) concerned by the procedure or to the medicinal product(s) of the represented group, the response should be stated as “not applicable” with a short explanation.
It should be noted that the responsibility for the quality of the submitted documentation lies with the MAHs and is crucial to the overall assessment. The data presented in the submissions should be intended exclusively for the purposes of the concerned procedure. The information and data contained in the individual submissions will be assessed and reflected in the assessment reports related to the concerned procedure. As a general rule, such information and data will not be redacted from the assessment reports with respect to individual products prior to sharing them with all concerned MAHs. Indeed for transparency reasons and in order to respect the right of defence of the MAHs concerned by the procedure, the Agency will share all the information and data relevant for the scientific assessment with all concerned MAHs. Moreover in general, it is not expected that individual submissions by the MAHs will include commercially confidential information.
It should be noted that neither the Agency nor the MAHs can use the information and data contained in the submissions for any other purposes than those related to the concerned procedure.
All submissions are expected to be submitted in English and electronically only (please refer to Question 19). Submission of responses concerning the Article 31 Pharmacovigilance referral with regards to centrally authorised products (CAPs) should follow the requirements for post-authorisation procedures for CAPs (e.g. submission via e-CTD).
In case MAHs formed a group (please refer to Question 11), the cover letter accompanying the single consolidated response and/or request for oral explanation should clearly identify the parties responsible for the submission/request.
Responses from the marketing authorisation holders (MAHs) should be submitted within the timeline specified on the procedure page.
All submissions for referral procedures should be sent via the eSubmission Gateway or eSubmission Web Client. These portals send automated acknowledgement of receipt of submission, or of failed submission if an error occurred. Responses for nationally authorised products (NAPs) and for centrally authorised products (CAPs) submitted via these portals are available in the common repository and will be considered delivered to all Committee members and alternates.
The Agency no longer accepts electronic submissions for referrals on CD or DVD. Additional copies of submissions should not be sent directly to the NCAs on CD/DVD or via common European submission portal (CESP) as this might cause delays in processing the submissions.
For advanced therapy medicinal product (ATMP), additional submission requirements apply. Please refer to the Dossier requirements for centrally authorised products.
The Agency strongly recommends using the electronic Common Technical Document (eCTD) or NeeS (Non-eCTD electronic Submission) formats for submissions related to referrals. For referral submissions related to CAPs, it is mandatory to use the eCTD format. It is not possible to submit a standalone joint eCTD for the CAPs concerned. Responses should always be submitted individually as the next sequence in each CAP product lifecycle. However for submissions related to NAPs, if the documentation is common to several medicinal products and/or MAHs, a single joint eCTD should be submitted. Additional copies of the same eCTD should not be submitted.
For all types of submissions, responses should be presented in the modular format.
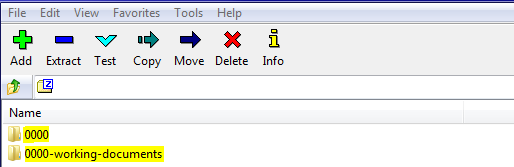
Recommended folder structure:
Documentation can be included in respective modules following the CTD location as referenced in the recommended folder structure, further, root folder should be 4 digits (between 0000-9999), e.g. Submission 0000 as below:

Any working documents (for example: documents in Word format) should be outside the root submission folder, e.g. as following:
Information on the required naming conventions and file formats can be found in detailed examples of filenames for different application types and in the eSubmission Gateway Web Client - Guidance for Applicants. For more information please refer to eSubmission website.
There is no need to send separate paper cover letters for these submissions, as the cover letter will be in the relevant part of eCTD module 1 in PDF format.
Should you have any questions regarding your submission, please contact us via email: AskEMA , for any technical issues visit the EMA Service Desk portal.
References:
Submissions from the marketing authorisation holder(s) (MAHs) concerned by the procedure are directly available in the common repository to the Pharmacovigilance Risk Assessment Committee (PRAC) (co-)rapporteurs to be considered for the assessment.
All information gathered will be assessed within an agreed timeframe as published on the Article 31 pharmacovigilance referral page. The assessment report(s) prepared by the PRAC (co-)rapporteur will reflect all data submitted and considered for the review.
The PRAC (co-)rapporteur’s assessment report(s) will be circulated to the PRAC members for comments. These will also be shared with the Committee for Medicinal Products for Human Use (CHMP) (co-)rapporteurs (in case at least one centrally authorised product is included in the scope of the procedure) or with the Co-ordination Group for Mutual Recognition and Decentralised Procedures (CMDh) member with a leading role (in case the concerned products are only nationally authorised including via the mutual recognition and decentralised procedures) for comments, as applicable (please refer to Question 16).
Please note that the timelines provided below are for guidance purposes only and refer to active days, which correspond to the time the Pharmacovigilance Risk Assessment Committee (PRAC) takes to assess the data provided.
The timelines following a 30 days assessment period are as follows:
|
Article 31 pharmacovigilance referral – Timetable for the assessment |
Active day |
|
Notification of a referral to the PRAC/Agency secretariat |
Day 0
|
|
Discussion at the first meeting of the PRAC following receipt of the notification:
|
Day 1 |
|
Preparation and submission of written explanations by the MAH(s) in response to the PRAC list of questions |
Clock Stop |
|
Re-start of the procedure following submission of the responses in accordance with published submission dates |
Clock re-start
|
|
Circulation of the PRAC (co-)rapporteur's assessment report(s) on the MAH(s)' written responses |
Day 20 |
|
Comments in writing from PRAC members on the (co-) rapporteur's assessment reports |
Day 25 |
|
Discussion at the PRAC meeting:
|
Day 30 |
The dates to be followed in accordance with the above timetable by the PRAC for each month can be found in Timetable: Safety referral (Article-20 and Article-31 pharmacovigilance)
The PRAC may extend the time limit up to 150 active days to allow for the assessment of further data provided as answers to the PRAC list of outstanding issues, oral explanation, public (and non-public) hearing and/or in case the PRAC requires input from a scientific advisory group or from an ad-hoc expert group to support the PRAC recommendation.
As a general rule, a clock-stop of up to one month will apply. For an extension of the clock-stop adopted by the PRAC, the MAH should send a justified request to the Agency for agreement by the PRAC. The letter specifying the length of the requested extension should be addressed to the PRAC Chairperson, signed and sent electronically to the EMA procedure manager. In preparing the justification, the MAH should consider the seriousness and urgency of the issue under consideration and the impact the extension may have on public health. The PRAC will consider the request, and if agreed, an extended timetable will be adopted. All MAHs involved in the procedure, will be informed of the PRAC outcome.
The PRAC assessment of responses to the list of outstanding issues will take up to 30 or, in exceptional cases, 60 days depending on the complexity and amount of data provided by the MAH(s).
All marketing authorisation holder(s) with medicinal products included in the scope of the Article 31 pharmacovigilance referral will be provided with the Pharmacovigilance Risk Assessment Committee (PRAC) (co-) rapporteur's assessment report(s) electronically via email/Eudralink.
The Pharmacovigilance Risk Assessment Committee (PRAC) may decide that there are issues that need to be addressed orally by the marketing authorisation holder(s) (MAHs). In such a case, the MAH(s) will be duly informed in advance of the issues to be addressed during the oral explanation.
The MAH(s) may also request to the PRAC to present their views in an oral explanation. In such a case, the MAH(s) should send to the EMA procedure manager a request addressed to the PRAC stating the reasons(s) and specifying the issue(s) to be addressed during the oral explanation. The PRAC will take due account of the request and will decide whether the oral explanation will be held.
Oral explanation(s) should take place during the assessment phase and after the receipt of the PRAC (co-)rapporteur’s assessment report(s). Further detailed information on organisational aspects of the oral explanation can be found in .
The MAH(s) can provide the oral explanation on their own behalf or on behalf of a group of MAHs.
Where the urgency of the matter permits, the PRAC may also hold public hearings, on justified grounds, particularly with regard to the extent and seriousness of the safety concern.
When the PRAC is of the opinion that a public hearing should be convened, the hearing shall be held in accordance with the modalities and rules specified by the Agency and will be announced on the Agency’s website. The announcement will also specify the modalities of participation. Further information can be found in Public hearings.
In case the PRAC has decided to hold a public hearing, a MAH or another person intending to submit confidential data relevant to the subject matter of the procedure may request permission to present that data to the PRAC in a non-public hearing. Such request should be duly justified on the grounds of confidentiality of the data to be presented. A non-public hearing can only be held whenever the PRAC has decided to hold a public hearing.
If the marketing authorisation (MA) for a concerned product is transferred during the referral, both the former and the new marketing authorisation holder (MAH) should update the Article 57 database without delay.
For nationally authorised product (including via mutual recognition and decentralised procedures)The new MAH should provide a copy of the transfer decision of the relevant competent authority and, if relevant, information on the new contact person, to the EMA procedure manager (please refer to Question 10).
Following receipt of the transfer decision, the Agency will inform the former MAH that they are no longer included in the Article 31 pharmacovigilance referral procedure, in relation to the MA transferred.
For a centrally authorised product (CAP), if during the referral procedure the MA is transferred, the former MAH should inform the EMA procedure manager and the appropriate procedure should be followed (please refer to Transfer of marketing authorisation: questions and answers).
The product listing published at the start of the procedure (please refer to Question 8) will be updated accordingly and republished on the Agency’s website on the specific procedure page.
If during the referral procedure, the name of a nationally authorised product (including via mutual recognition and decentralised procedures) changes or, if the name and/or address of the marketing authorisation holder (MAH) changes or, if the marketing authorisation (MA) is withdrawn, the MAH(s) should update the Article 57 database without delay and inform the EMA procedure manager.
For nationally authorised product (including via mutual recognition and decentralised procedures), following official confirmation of the change, the Agency will inform the MAH/applicant that the change has been noted. If the Article 57 database is updated within 30 days of the start of the procedure, these changes will be included in the revised product listing that will be published at day 30. After day 30 the product listing will not be subject to any changes except in case of MA transfer (please refer to Question 24).
If the contact person changes, the MAH should also inform the EMA procedure manager and, if the contact person is the QPPV, update the Article 57 database without delay.
For a centrally authorised product (CAP), if during the referral procedure, the name of the product or the name and/or address of the MAH changes or, if the contact person changes or, if the MA is withdrawn, the MAH should inform the EMA procedure manager and the appropriate procedure should be followed (please refer to Changing the (invented) name of a centrally authorised medicine: questions and answers ,Other post-authorisation activities: questions and answers and Withdrawn-product notification: questions and answers - MAH notification time).
The Pharmacovigilance Risk Assessment Committee (PRAC) will issue a recommendation on the safety matter referred under Article 31 within 60 days of the start date of the procedure. The PRAC may extend that period up to 150 days, to take into account the views of the marketing authorisation holder(s), in case a public (and non-public) hearing is held and/or an ad-hoc expert/scientific advisory group (SAG) meeting is needed.
The PRAC recommendation will usually be adopted on the last day of the PRAC plenary meeting.
The Pharmacovigilance Risk Assessment Committee (PRAC) recommendation on the safety issue referred under Article 31 shall include any or a combination of the following:
Where the recommendation is for the MA(s) to be varied, including changes to the information in the summary of product characteristics (SmPC), labelling and/or package leaflet (PL), the recommendation will include the suggested wording of such amendments.
With regards to point (b), the recommendation will specify any conditions or restrictions to which the MA should be made subject. Conditions for the safe and effective use of the product(s) can include, but are not limited to, requesting the marketing authorisation holder(s) to conduct a post-authorisation safety study and/or to implement additional risk minimisation measures.
The PRAC recommendation can be adopted by consensus or by majority vote. In the event of adoption by majority, the divergent positions of the concerned PRAC members and the grounds on which they are based will be appended to the recommendation issued by the PRAC.
References:
The Pharmacovigilance Risk Assessment Committee (PRAC) recommendation will include:
The outcome of the Pharmacovigilance Risk Assessment Committee (PRAC) recommendation will be included in the PRAC meeting highlights that are released on the next working day following the PRAC plenary meeting together with a summary of the PRAC recommendation.
The PRAC assessment report detailing the PRAC recommendation will be published on the procedure page around one week following the adoption of the European Commission Decision or Co-ordination Group for Mutual Recognition and Decentralised Procedures (CMDh) consensus position as applicable (please refer to Question 39).
Reference:
The marketing authorisation holder(s) (MAHs) of medicinal products concerned and identified at the start of the procedure will receive the Pharmacovigilance Risk Assessment Committee (PRAC) recommendation electronically via email/Eudralink during the week following the PRAC meeting when the recommendation was adopted.
When no request for re-examination of the Pharmacovigilance Risk Assessment Committee (PRAC) recommendation has been received 15 calendar days following its receipt by the MAH(s), the PRAC recommendation is considered final.
When a re-examination procedure is initiated, the PRAC will adopt a final recommendation and within 60 calendar days of receipt of the detailed grounds for re-examination (please refer to Question 31).
The final PRAC recommendation is sent to:
The CHMP or CMDh will consider the PRAC recommendation at their following plenary meeting and will agree on the timeframe needed to issue an opinion or position, respectively. This timeframe should not exceed 30 days after receipt of the PRAC recommendation (please refer to Question 33).
In parallel, some of the concerned MAHs, will be contacted by the Agency with a proposal for worksharing of the translation process in all EU official languages (please refer to Question 38).
Following the receipt of the final Pharmacovigilance Risk Assessment Committee (PRAC) recommendation, the Committee for Medicinal Products for Human Use (CHMP) or Co-ordination Group for Mutual Recognition and Decentralised Procedures (CMDh) will consider it at their next plenary meeting. As a general rule, the aim will be to adopt the CHMP opinion or CMDh position at the first plenary meeting following the receipt of the PRAC recommendation.
However in some cases, the CHMP or CMDh may agree on the need to further consider the PRAC recommendation. In such cases, the CHMP opinion or CMDh position will be adopted within 30 days after receipt of the PRAC recommendation.
This decision will be reflected in the CHMP or CMDh meeting highlights published on the next working day following the plenary meetings.
The overall process including timelines for adoption of an opinion/position is the following:
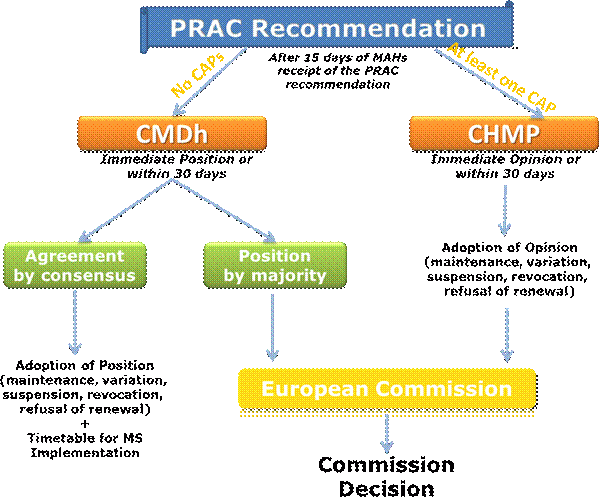
The Committee for Medicinal Products for Human Use (CHMP) or Co-ordination Group for Mutual Recognition and Decentralised Procedures (CMDh) will consider the Pharmacovigilance Risk Assessment Committee (PRAC) recommendation and assessment report, and will adopt by consensus or by majority vote, a CHMP opinion or CMDh position on the maintenance, variation, suspension or revocation of the marketing authorisations (MAs) concerned (please refer to Question 27).
Exceptionally, an oral explanation may be held in front of the CHMP/CMDh. The CHMP/CMDh decides whether the oral explanation will be held.
Where the CHMP opinion or CMDh position differs from the recommendation of the PRAC, the CHMP or CMDh will attach to its opinion or position an explanation of the scientific grounds for the differences.
The Committee for Medicinal Products for Human Use (CHMP) opinion or Co-ordination Group for Mutual Recognition and Decentralised Procedures (CMDh) position will include:
An EMA public health communication (including a summary of the CHMP opinion/CMDh position and targeted information for healthcare professional and patients) and, if applicable, the wording changes to be applied to the product information, and the timetable for implementation of the CMDh position by consensus, will be published on the Friday of the plenary meeting week. In addition, a brief outcome of the Committee for Medicinal Products for Human Use (CHMP) opinion or Co-ordination Group for Mutual Recognition and Decentralised Procedures (CMDh) position, as applicable, will be included in the CHMP meeting highlights/ CMDh report that are released following the plenary meetings. .
The CHMP opinion or CMDh position will be published on the procedure page following the adoption of the European Commission Decision or CMDh consensus position as applicable (please refer to Question 39).
Reference:
The marketing authorisation holder(s) of medicinal products concerned and identified at the start of the procedure, will receive the Committee for Medicinal Products for Human Use (CHMP) opinion or Co-ordination Group for Mutual Recognition and Decentralised Procedures (CMDh) position electronically via email/Eudralink during the week following the CHMP or CMDh plenary meetings during which the CHMP opinion or CMDh position was adopted.
The marketing authorisation holder(s) (MAHs) of medicinal products authorised nationally (including via the mutual recognition or decentralised procedures) will have to provide translations in all EU languages (including Icelandic and Norwegian, if applicable1) of the following annexes to the Committee for Medicinal Products for Human Use (CHMP) opinion or Co-ordination Group for Mutual Recognition and Decentralised Procedures (CMDh) position:
Only one translation per EU language is required, therefore the MAHs actively involved in the procedure will be presented with a proposal for worksharing for the translation process. Not all MAHs may be involved in the translation process. MAHs that have not been contacted to participate in the worksharing process will be provided the set of translations at a later stage.
The Agency will contact the MAHs as early as possible to ensure the smooth running of the worksharing process. The translations will have to be provided to the Member States contact points for linguistic check by Day +5 (i.e. 5 days after adoption of the opinion or the position) and copied to the Agency. Member States may send linguistic comments until Day +19. The MAH should send the translations amended accordingly together with the completed Practical information on translations for referral procedures (human) to the Agency by Day +22.
The MAH(s) of centrally authorised products involved in the procedure will have to provide the full product information in all EU languages within the same timeframe (i.e. 5 days after adoption of the opinion) to the MSs contact points for linguistic check and copied to the Agency.
Detailed information on the translation process of the CHMP opinion or CMDh position can be found in Practical information on translations for referral procedures (human)
1 If authorised in Iceland and Norway
In case of a Committee for Medicinal Products for Human Use (CHMP) opinion or Co-ordination Group for Mutual Recognition and Decentralised Procedures (CMDh) position by majority vote, the Agency together with the concerned marketing authorisation holder(s) (MAHs) and national competent authorities (NCAs) in the Member States (MSs) will finalise the translations and send these to the European Commission (EC).
The EC will then start the decision-making process leading to the adoption of a binding decision addressed either to the MAHs or to MSs and notified to the MAHs, depending on whether the decision concerns centrally authorised products (CAPs) or nationally authorised products (including via the mutual recognition and decentralised procedures), respectively.
In case of CMDh position by consensus, the Agency together with the concerned MAHs and the NCAs in the MSs will finalise the translations. The consensus position will be implemented by the MSs in accordance with the timetable determined in the position (please refer to Question 33).
Detailed information on the decision-making process can be found here.
The MAHs of CAPs need to submit an eCTD closing sequence with the final documents within 5 days following the EC decision.
For NAPs, the CMDh recommendation for implementation of Commission Decisions or CMDh position can be found here.
References
The PRAC assessment report will be published on the procedure page, in English only, around one week following the adoption of the European Commission (EC) decision or Co-ordination Group for Mutual Recognition and Decentralised Procedures (CMDh) consensus position. Within four weeks of the adoption of the EC decision or eight weeks of the CMDh consensus position, the Committee for Medicinal Products for Human Use (CHMP) Opinion or CMDh position (as applicable) with its annexes in all EU languages will be published on the procedure page. The page will also be updated to reflect the date of the EC decision or the CMDh consensus position, as applicable.
Reference: