Meeting highlights from the Committee for Medicinal Products for Human Use (CHMP) 19-22 April 2021
NewsHumanGeneric and hybrid medicinesMedicinesReferrals
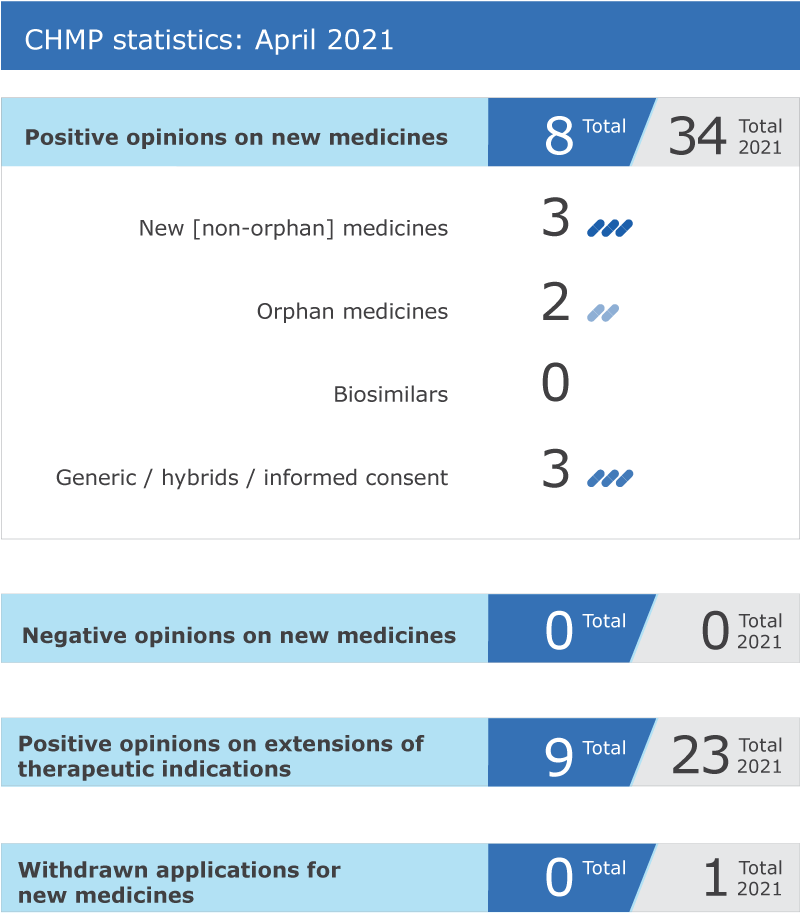
EMA’s human medicines committee (CHMP) recommended eight medicines for approval at its April 2021 meeting.
The Committee gave a positive opinion for Enspryng* (satralizumab) for the treatment of neuromyelitis optica spectrum disorders (NMOSD) in patients from 12 years of age who are anti-aquaporin-4 IgG (AQP4-IgG) seropositive. See more details in the news announcement in the grid below.
Evkeeza (evinacumab) was granted a positive opinion by the Committee for the treatment of adult and adolescent patients aged 12 years and older with homozygous familial hypercholesterolemia (HoFH). Evkeeza was reviewed under EMA’s accelerated assessment programme.
The Committee recommended granting a marketing authorisation for Adtralza (tralokinumab) for the treatment of adults with moderate to severe atopic dermatitis who are candidates for systemic therapy.
Koselugo* (selumetinib) was granted a positive opinion for the treatment of paediatric patients with neurofibromatosis type 1 (NF1) plexiform neurofibromas (PN).
The Committee adopted a positive opinion for Onureg (azacitidine) for the maintenance treatment of patients with acute myeloid leukemia.
The CHMP recommended granting marketing authorisations for two generic medicines: Abiraterone Krka (abiraterone acetate) for the treatment of adult men with metastatic prostate cancer and Celsunax (ioflupane (123I)) intended for detecting loss of functional dopaminergic neuron terminals in the striatum.
The Committee recommended granting a marketing authorisation for the hybrid medicine Jayempi (azathioprine) intended for prophylaxis against transplant rejection and as immunosuppressive antimetabolite either alone or in combination with other agents to influence the immune response in a variety of diseases. Hybrid applications rely in part on the results of pre-clinical tests and clinical trials of an already authorised reference product and in part on new data.
The Committee recommended extensions of indication for Aubagio, BiResp Spiromax, DuoResp Spiromax, Maviret, Nulojix, Opdivo, Tagrisso, Venclyxto and Yervoy.
The agenda of the April 2021 CHMP meeting is published on EMA's website. Minutes of the March 2021 CHMP meeting will be published in the coming weeks.
Key figures from the April 2021 CHMP meeting are represented in the graphic below.
*This product was designated as an orphan medicine during its development. Orphan designations are reviewed by EMA's Committee for Orphan Medicinal Products (COMP) at the time of approval to determine whether the information available to date allows maintaining the medicine’s orphan status and granting the medicine ten years of market exclusivity.
| Name of medicine | Adtralza |
| International non-proprietary name (INN) | tralokinumab |
| Marketing-authorisation applicant | Leo Pharma A/S |
| Therapeutic indication | Treatment of moderate?to?severe atopic dermatitis |
| More information | Adtralza: Pending EC decision |
| Name of medicine | Enspryng |
| INN | satralizumab |
| Marketing-authorisation applicant | Roche Registration GmbH |
| Therapeutic indication | Treatment of neuromyelitis optica spectrum disorders (NMOSD) |
| More information |
Press release: New treatment for rare autoimmune disease of nerve cells |
| Name of medicine | Evkeeza |
| INN | evinacumab |
| Marketing-authorisation applicant | Regeneron Ireland Designated Activity Company (DAC) |
| Therapeutic indication | Treatment of adult and adolescent patients with homozygous familial hypercholesterolaemia (HoFH) |
| More information | Evkeeza: Pending EC decision |
| Name of medicine | Koselugo |
| INN | selumetinib |
| Marketing-authorisation applicant | AstraZeneca AB |
| Therapeutic indication | Treatment of paediatric patients with neurofibromatosis type 1 (NF1) plexiform neurofibromas (PN) |
| More information | Koselugo: Pending EC decision |
| Name of medicine | Onureg |
| INN | azacitidine |
| Marketing-authorisation applicant | Celgene Europe BV |
| Therapeutic indication | Maintenance treatment of patients with acute myeloid leukemia |
| More information | Onureg: Pending EC decision |
| Name of medicine | Abiraterone Krka |
| International non-proprietary name (INN) | abiraterone acetate |
| Marketing-authorisation applicant | KRKA, d.d., Novo mesto |
| Therapeutic indication | Treatment of metastatic prostate cancer |
| More information | Abiraterone Krka: Pending EC decision |
| Name of medicine | Celsunax |
| INN | ioflupane (123I) |
| Marketing-authorisation applicant | Pinax Pharma GmbH |
| Therapeutic indication | Detecting loss of functional dopaminergic neuron terminals in the striatum |
| More information | Celsunax: Pending EC decision |
| Name of medicine | Jayempi |
| INN | azathioprine |
| Marketing-authorisation applicant | Nova Laboratories Ireland Limited |
| Therapeutic indication | Prophylaxis against transplant rejection and as immunosuppressive antimetabolite |
| More information | Jayempi: Pending EC decision |
| Name of medicine | Aubagio |
| INN | teriflunomide |
| Marketing-authorisation holder | Sanofi-Aventis Groupe |
| More information | Aubagio: Pending EC decision |
| Name of medicine | BiResp Spiromax |
| INN | budesonide / formoterol |
| Marketing-authorisation holder | Teva Pharma B.V. |
| More information | BiResp Spiromax: Pending EC decision |
| Name of medicine | DuoResp Spiromax |
| INN | budesonide / formoterol |
| Marketing-authorisation holder | Teva Pharma B.V. |
| More information | DuoResp Spiromax: Pending EC decision |
| Name of medicine | Maviret |
| INN | glecaprevir / pibrentasvir |
| Marketing-authorisation holder | AbbVie Deutschland GmbH & Co. KG |
| More information | Maviret: Pending EC decision |
| Name of medicine | Nulojix |
| INN | belatacept |
| Marketing-authorisation holder | Bristol-Myers Squibb Pharma EEIG |
| More information | Nulojix: Pending EC decision |
| Name of medicine | Opdivo |
| INN | nivolumab |
| Marketing-authorisation holder | Bristol-Myers Squibb Pharma EEIG |
| More information | Opdivo: Pending EC decision |
| Name of medicine | Tagrisso |
| INN | osimertinib |
| Marketing-authorisation holder |
AstraZeneca AB |
| More information | Tagrisso: Pending EC decision |
| Name of medicine | Venclyxto |
| INN | venetoclax |
| Marketing-authorisation holder | AbbVie Deutschland GmbH & Co. KG |
| More information | Venclyxto: Pending EC decision |
| Name of medicine | Yervoy |
| INN | ipilimumab |
| Marketing-authorisation holder | Bristol-Myers Squibb Pharma EEIG |
| More information | Yervoy: Pending EC decision |