Meeting highlights from the Committee for Medicinal Products for Human Use (CHMP) 22-25 June 2015
NewsHuman
Ten new medicines, including two enzyme replacement therapies for rare diseases, recommended for approval
Ten new medicines have been recommended for approval at the June 2015 meeting of the Committee for Medicinal Products for Human Use (CHMP).
Two enzyme replacement therapies for the treatment of rare genetic diseases received a positive opinion from the CHMP: Kanuma (sebelipase alfa) for the treatment of lysosomal acid lipase deficiency, and Strensiq (asfotase alfa), the first therapy for the bone disease hypophosphatasia that started in childhood. For more information on these two enzyme replacement therapies, both of which have an orphan designation, please see the press releases in the grid below.
The CHMP also recommended granting a marketing authorisation for Farydak (panobinostat) for the treatment of relapsed and/or refractory multiple myeloma. Farydak is the first cancer medicine that targets enzymes known as histone deacetylases. Farydak has an orphan designation. For more information on Farydak, please see the press release in the grid below.
Odomzo (sonidegib) received a position opinion from the CHMP for the treatment of basal cell carcinoma.
The CHMP also recommended Respreeza (human alpha1-proteinase inhibitor) for the treatment of alpha1-proteinase inhibitor deficiency.
Three generic medicines received positive opinions from the CHMP: Docetaxel Hospira UK Limited (docetaxel) for the treatment of breast cancer, non-small cell lung cancer, prostate cancer, gastric adenocarcinoma, and head and neck cancer, Duloxetine Zentiva (duloxetine) for the treatment of major depressive disorder, diabetic peripheral neuropathic pain and generalised anxiety disorder and Pregabalin Accord (pregabalin) for the treatment of epilepsy and generalised anxiety disorder.
Two 'hybrid' medicines received positive opinions from the Committee: Aripiprazole Sandoz (aripiprazole) for the treatment of schizophrenia and the prevention and treatment of manic episodes in bipolar 1 disorder, and Raxone (idebenone) for the treatment of visual impairment in patients with Leber's hereditary optic neuropathy. Raxone has an orphan designation. Hybrid applications rely in part on the results of pre-clinical tests and clinical trials carried out with the reference product and in part on new data.
Negative opinion on new medicine
The CHMP adopted a negative opinion recommending the refusal of a marketing authorisation for the advanced therapy medicinal product Heparesc (human heterologous liver cells). Heparesc was intended to be used for the treatment of certain urea cycle disorders.
Four recommendations on extensions of therapeutic indication
The Committee recommended extensions of indication for Humira, Levemir, Perjeta and Voncento. For more information on the extension of indication for Humira, please see the press release in the grid below.
Outcome of review on adrenaline auto-injectors
The CHMP recommended several measures, including the introduction of educational material, to ensure that patients and carers use adrenaline auto-injectors successfully. An adrenaline auto-injectors are potentially life-saving treatments for anaphylaxis (severe allergic reactions) while the patient waits for emergency medical assistance. For more information on the outcome of this review, please see the public health communication in the grid below.
Agenda and minutes
The agenda of the June 2015 meeting is published on EMA's website. The minutes of the meeting will be published during the week following the July CHMP meeting.
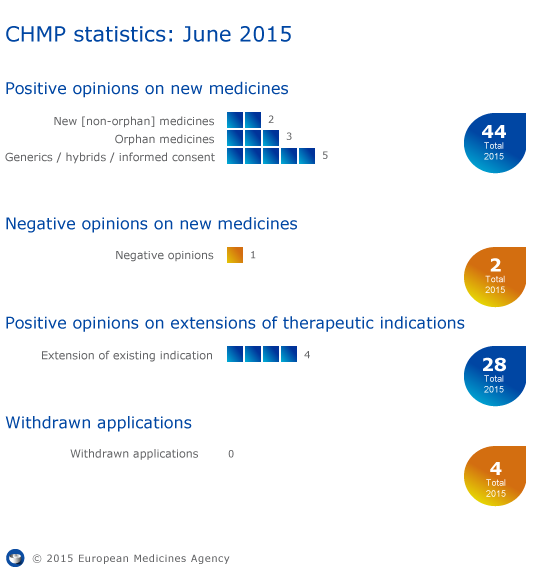
CHMP statistics
Key figures from the June 2015 CHMP meeting are represented in the graphic below.
More information on this, and all other outcomes of the CHMP's June 2015 meeting, is available in the grid below.
| Name of medicine | Farydak |
|---|---|
| International non-proprietary name (INN) | panobinostat |
| Marketing-authorisation applicant | Novartis Europharm Ltd |
| Therapeutic indication | Treatment of multiple myeloma |
| More information |
CHMP summary of positive opinion for Farydak Press release: First HDAC inhibitor for treatment of multiple myeloma recommended for approval in EU |
| Name of medicine | Kanuma |
|---|---|
| INN | sebelipase alfa |
| Marketing-authorisation applicant | Synageva BioPharma Ltd |
| Therapeutic indication | Treatment of lysosomal acid lipase deficiency |
| More information | CHMP summary of opinion for Kanuma
Press release: EMA fast-tracks enzyme replacement therapy for lysosomal acid lipase deficiency |
| Name of medicine | Odomzo |
|---|---|
| INN | sonidegib |
| Marketing-authorisation applicant | Novartis Europharm Ltd |
| Therapeutic indication | Treatment of basal cell carcinoma |
| More information | CHMP summary of positive opinion for Odomzo |
| Name of medicine | Respreeza |
|---|---|
| INN | human alpha1-proteinase inhibitor |
| Marketing-authorisation applicant | CSL Behring GmbH |
| Therapeutic indication | Treatment of alpha1-proteinase inhibitor deficiency |
| More information | CHMP summary of positive opinion for Respreeza |
| Name of medicine | Strensiq |
|---|---|
| INN | asfotase alfa |
| Marketing-authorisation applicant | Alexion Europe SAS |
| Therapeutic indication | Treatment of paediatric-onset hypophosphatasia |
| More information |
CHMP summary of positive opinion for Strensiq
Press release: First treatment recommended for rare bone disease |
| Name of medicine | Docetaxel Hospira UK Limited |
|---|---|
| INN | docetaxel |
| Marketing-authorisation applicant | Hospira UK Limited |
| Therapeutic indication | Treatment of breast cancer, non small cell lung cancer, prostate cancer, gastric adenocarcinoma, and head and neck cancer |
| More information |
| Name of medicine | Duloxetine Zentiva |
|---|---|
| INN | duloxetine |
| Marketing-authorisation applicant | Zentiva, k.s. |
| Therapeutic indication | Treatment of major depressive disorder, diabetic peripheral neuropathic pain and generalised anxiety disorder |
| More information | CHMP summary of positive opinion for Duloxetine Zentiva |
| Name of medicine | Pregabalin Accord |
|---|---|
| INN | pregabalin |
| Marketing-authorisation applicant | Accord Healthcare Limited |
| Therapeutic indication | Treatment of epilepsy and generalised anxiety disorder |
| More information | CHMP summary of positive opinion for Pregabalin Accord |
| Name of medicine | Aripiprazole Sandoz |
|---|---|
| INN | aripiprazole |
| Marketing-authorisation applicant | Sandoz GmbH |
| Therapeutic indication | Treatment of schizophrenia and treatment and prevention of manic episodes in bipolar I disorder |
| More information | CHMP summary of positive opinion for Aripiprazole Sandoz |
| Name of medicine | Raxone |
|---|---|
| INN | idebenone |
| Marketing-authorisation applicant | Santhera Pharmaceuticals (Deutschland) GmbH |
| Therapeutic indication | Treatment of Leber's Hereditary Optic Neuropathy |
| More information | CHMP summary of positive opinion for Raxone |
| Name of medicine | Heparesc |
|---|---|
| INN | human heterologous liver cells |
| More information | Questions and answers on refusal of the marketing authorisation for Heparesc (human heterologous liver cells) |
| Name of medicine | Humira |
|---|---|
| INN | adalimumab |
| Marketing-authorisation holder | AbbVie Ltd |
| More information |
CHMP post-authorisation summary of positive opinion for Humira
Press release: First medicine recommended for approval for hidradenitis suppurativa |
| Name of medicine | Levemir |
|---|---|
| INN | insulin detemir |
| Marketing-authorisation holder | Novo Nordisk A/S |
| More information | CHMP post-authorisation summary of positive opinion for Levemir |
| Name of medicine | Perjeta |
|---|---|
| INN | pertuzumab |
| Marketing-authorisation holder | Roche Registration Ltd |
| More information | CHMP post-authorisation summary of positive opinion for Perjeta |
| Name of medicine | Voncento |
|---|---|
| INN | human coagulation factor viii / human von willebrand factor |
| Marketing-authorisation holder | CSL Behring GmbH |
| More information | CHMP post-authorisation summary of positive opinion for Voncento |
| Name of medicine | Adrenaline auto-injectors |
|---|---|
| INN | adrenaline (epinephrine) |
| More information | Better training tools recommended to support patients using adrenaline auto-injectors |
| Name of medicine | Amoxil |
|---|---|
| INN | amoxicillin |
| Marketing-authorisation holder | GlaxoSmithKline |
| More information | Questions and answers on Amoxil |